- Navi Mumbai, Maharashtra, India, 400705.
- +91 98200 02282
- sales@gmchemical.com
Polyvinyl Acetate Phthalate
- Home
- Polyvinyl Acetate Phthalate
Get in Touch
If you have any queries, please feel free to fill out the form below, and we will get back to you at the earliest.
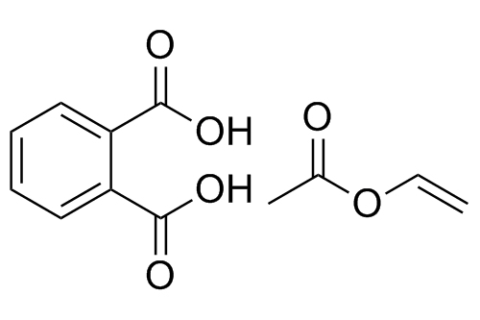
Polyvinyl Acetate Phthalate (PVAP)
Nonproprietary Names:
Polyvinyl Acetate Phthalate
Synonyms:
Polyvinyl Acetate Phthalate, PVAP
Chemical Name & CAS No.
Polyvinyl Acetate Phthalate; [34481-48-6]
Functional Category:
Coating Agent.
Product Description:
Polyvinyl acetate phthalate is a reaction product of phthalic anhydride, sodium acetate, and a partially hydrolyzed polyvinyl alcohol. The polyvinyl alcohol is a low molecular weight grade, and 87–89 mole percent is hydrolyzed. Therefore, the polyvinyl acetate phthalate polymer is a partial esterification of a partially hydrolyzed polyvinyl acetate. Polyvinyl acetate phthalate is a free-flowing white to off-white powder and may have a slight odor of acetic acid. The material is essentially amorphous.
Polyvinyl acetate phthalate is a viscosity-modifying agent that is used in pharmaceutical formulations to produce enteric coatings for products and for the core sealing of tablets prior to a sugar-coating process. Polyvinyl acetate phthalate does not exhibit tackiness during coating and produces strong robust films.
Solubility of Polyvinyl Acetate Phthalate
Soluble in ethanol and methanol; sparingly soluble in acetone and propan-2-ol; practically insoluble in chloroform, dichloromethane, and water. In buffer solutions, polyvinyl acetate phthalate (200 mg/L) is insoluble below pH 5 and becomes soluble at pH values above 5. Polyvinyl acetate phthalate shows a sharp solubility response with pH; this occurs at pH 4.5–5.0, which is lower than for most other polymers used for enteric coatings. Solubility is also influenced by ionic strength.
Stability and Handling:
Polyvinyl acetate phthalate should be stored in airtight containers. It is relatively stable to temperature and humidity, and does not age, giving predictable release profiles even after prolonged storage. At high temperature and humidity, polyvinyl acetate phthalate undergoes less hydrolysis. Polyvinyl acetate phthalate is used in oral pharmaceutical formulations and is generally regarded as an essentially nonirritant and nontoxic material when used as an excipient. Observe normal precautions appropriate to the circumstances and quantity of material handled. Gloves and eye protection are recommended.
Specification of Polyvinyl Acetate Phthalate:
| Properties | Specifications |
| USP / NF | |
| Identification | A. The Sample solution in the Assay exhibits a maximum at 277 ± 3 nm. B. The solution shows a vivid green fluorescence. C. A film is deposited as the methanol evaporates. |
| Viscosity @ 25°C | 7cps – 11cps |
| Water Content | NMT 5.0% |
| Residue on Ignition | NMT 1.0% |
| Free Phthalic Acid | NMT 0.6% |
| Free Acid Other than Phthalic | NMT 0.6% |
| Phthalyl Content | 55.0% – 62.0% |
-
Find Our Location
-
Call for Enquiries
-
Send Us a Message
About Us
G.M.Chemical started its operations in 1981 and has gradually emerged as not only the leading provider of Cellulose Acetate Phthalate [BP/USP/BPC/IP] to the Indian Pharmaceutical Industry but also to prestigious clients across the globe.
Quick Links
Products Link
Contact Us
- C-233/234 TTC Industrial Area, MIDC, Pawane, Navi Mumbai 400705, Maharashtra, India.
© 2026 G.M. Chemical. Designed by FQI. All rights reserved.