- Navi Mumbai, Maharashtra, India, 400705.
- +91 98200 02282
- sales@gmchemical.com
Cellulose Acetate
- Home
- Cellulose Acetate
Get in Touch
If you have any queries, please feel free to fill out the form below, and we will get back to you at the earliest.
Cellulose Acetate (CA)
Nonproprietary Names:
Cellulose Acetate (32% and 39.8% acetyl content)
Synonyms:
Acetic acid, cellulose ester; acetyl cellulose; cellulose diacetate; cellulosi acetas.
Chemical Name & CAS No.
Cellulose acetate [9004-35-7]
Functional Category:
Coating Agent, Extended-Release Agent, Sustain Release Agent, Taste Masking Agent, Semi permeable membrane for Osmotic (Controlled-release) drug delivery.
Product Description:
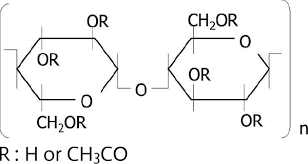
Cellulose acetate occurs as a hygroscopic white to off-white, free flowing powder, pellet, or flake. It is tasteless and odorless, or may have a slight odor of acetic acid. Cellulose acetate is cellulose in which a portion or all of the hydroxyl groups are acetylated. Cellulose acetate is available in a wide range of acetyl levels and chain lengths and thus molecular weights.
Cellulose acetate is widely used in pharmaceutical formulations both in sustained-release applications and for taste masking. Cellulose acetate is used as a semipermeable coating on tablets, especially on osmotic pump-type tablets and implants. This allows for controlled, extended release of actives. Cellulose acetate films, in conjunction with other materials, also offer sustained release without the necessity of drilling a hole in the coating as is typical with osmotic pump systems. Cellulose acetate and other cellulose esters have also been used to form drug-loaded micro particles with controlled-release characteristics. Cellulose acetate films are used in transdermal drug delivery systems and also as film coatings on tablets or granules for taste masking. For example, acetaminophen granules have been coated with a cellulose acetate-based coating before being processed to provide chewable tablets. Extended-release tablets can also be formulated with cellulose acetate as a directly compressible matrix former. The release profile can be modified by changing the ratio of active to cellulose acetate and by incorporation of plasticizer, but was shown to be insensitive to cellulose acetate molecular weight and particle size distribution. Therapeutically, cellulose acetate has been used to treat cerebral aneurysms, and also for spinal perimedullary arteriovenous fistulas.
Solubility of Cellulose Acetate
The solubility of cellulose acetate is greatly influenced by the level of acetyl groups present. In general, cellulose acetates are soluble in acetone–water blends of varying ratios, dichloro methane–ethanol blends, dimethyl formamide, and dioxane. The cellulose acetates of higher acetyl level are generally more limited in solvent choice than are the lower-acetyl materials.
Stability and Handling:
Cellulose acetate is stable if stored in a well-closed container in a cool, dry place. Cellulose acetate hydrolyzes slowly under prolonged adverse conditions such as high temperature and humidity, with a resultant increase in free acid content and odor of acetic acid. Cellulose acetate is widely used in oral pharmaceutical products and is generally regarded as a nontoxic and nonirritant material. Cellulose acetate is in compatible with strongly acidic or alkaline substances. Cellulose acetate is compatible with the following plasticizers: diethyl phthalate, polyethylene glycol, triacetin, and triethyl citrate. Observe normal precautions appropriate to the circumstances and quantity of material handled. Dust may be irritant to the eyes and eye protection should be worn. Like most organic materials in powder form, these materials are capable of creating dust explosions. Cellulose acetate is combustible.
Specification of Cellulose Acetate:
| Properties | Specifications | |||
|---|---|---|---|---|
| BP 2023 | USP 43 | EP 11.0 | IP 2022 | |
| Appearance | White, yellowish-white or greyish-white, hygroscopic powder or granules. | White powder or free flowing pellets. | White, yellowish-white or greyish-white, hygroscopic powder or granules | A white powder of free-flowing pellets, available in a range of viscosities and acetyl contents. |
| Solubility | Practically insoluble in water, soluble in acetone, in formic acid and in a mixture of equal volumes of methanol and methylene chloride, practically insoluble in ethanol | Soluble in methylene chloride, all acetyl content cellulose acetates soluble in dioxane and in dimethyl-formamide; insoluble in alcohol and in water | Practically insoluble in water, soluble in acetone, in formic acid and in a mixture of equal volumes of methanol and methylene chloride, practically insoluble in ethanol (96 per cent) | ———– |
| Identification | I.R. Spectra | I.R. Spectra | I.R. Spectra | I.R. Spectra |
| Viscosity | Only for information | ——– | Only for information | ———- |
| Acetyl Content (Labeled Acetyl Content) | 29.0% to 44.8% | 29.0% to 44.8% | 29.0% to 44.8% | 29.0% to 44.8% |
| Residue on Ignition / Sulfated Ash | NMT 0.1% | NMT 0.1% | NMT 0.1% | NMT 0.1% |
| Free Acid | NMT 0.1% | NMT 0.1% | NMT 0.1% | NMT 0.1% |
| Heavy metals | ——– | ——— | ——— | Max 20 ppm |
| Loss on drying | NMT 5.0% | NMT 5.0% | NMT 5.0% | NMT 5.0% |
| Microbial Contamination |
TAMC: 10³ CFU/g TYMC: 10² CFU/g Absence of Esch. coli Absence of Salmonella |
TAMC: 10³ CFU/g TYMC: 10² CFU/g Absence of Esch. coli Absence of Salmonella |
TAMC: 10³ CFU/g TYMC: 10² CFU/g Absence of Esch. coli Absence of Salmonella |
TAMC: 10³ CFU/g TYMC: 10² CFU/g Absence of Esch. coli Absence of Salmonella |
-
Find Our Location
-
Call for Enquiries
-
Send Us a Message
About Us
G.M.Chemical started its operations in 1981 and has gradually emerged as not only the leading provider of Cellulose Acetate Phthalate [BP/USP/BPC/IP] to the Indian Pharmaceutical Industry but also to prestigious clients across the globe.
Quick Links
Products Link
Contact Us
- C-233/234 TTC Industrial Area, MIDC, Pawane, Navi Mumbai 400705, Maharashtra, India.
© 2026 G.M. Chemical. Designed by FQI. All rights reserved.