- Navi Mumbai, Maharashtra, India, 400705.
- +91 98200 02282
- sales@gmchemical.com
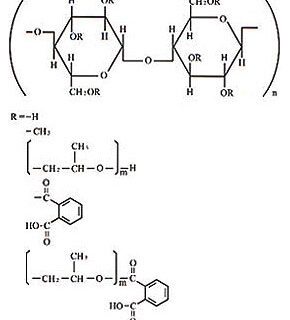
Hypromellose Phthalate
- Home
- Hypromellose Phthalate
Get in Touch
If you have any queries, please feel free to fill out the form below, and we will get back to you at the earliest.
Hypromellose Phthalate
Nonproprietary Names:
Hypromellose Phthalate
Synonyms:
HPMCP HydroxyPropyl MethylCellulose Phthalate or
Cellulose, 2-Hydroxypropyl methylether, phthalic acid ester.
Chemical Name & CAS No.
Hypromellose Phthalate; [9050-31-1]
Specifications:
| Item | HPP-55 | HPP-55S | HPP-50 |
| Dissolving pH | ≥ 5.5 | ≥ 5.0 | |
| Labeled Viscosity (cps) @ 20°C | 40 | 150 | 55 |
| Viscosity (cps) @ 20°C | 32 – 48 | 136 – 204 | 44 – 66 |
| Phthalyl Content | 27.0% – 35.0% | 21.0% – 27.0% | |
| Nominal Phthalyl Content | 31% | 31% | 24% |
| Description and Solubility | Conforms | ||
| Identification (Infrared Absorption) | Conforms | ||
| Water | No more than 5.0% | ||
| Residue on Ignition | No more than 0.20% | ||
| Chloride | No more than 0.07% | ||
| Heavy Metals | No more than 0.001% | ||
| Free Phthalic Acid | No more than 1.0% | ||
Physical form :
Powder/Flakes.
Properties :
“HPMCP” (hydroxypropyl methylcellulose phthalate), an enteric coating. It is widely used as an enteric coating agent by the pharmaceutical industry. “HPMCP”
It can dissolve at pH 5-5.5 and be controlled by varying the phthalyl content. Two types of different pH solubility, HP-55 and HP-50, are available. A suitable grade of “HPMCP” for a particular purpose should be selected in accordance with the properties and formulations.Moreover,HP-55S a special type of HP-55 which is distinguished by its higher molecular weight and higher resistance to simulated gastric fluid.
HP-55 is applicable as ageneral enteric coating agent.
HP-55S, because of its high degree of polymerisation compared with HP-55,tends to have higher viscosity solution,higher mechanical strenght of film and higher simulated gastric fluid resistance of the coating formulation.these characteristics are effective in reducing theamount of coating and in preventing crack generation in film applied to fragile tablets or granules.
HP-50 can be dissolved at a lower pH and is therefore applicable to preparation which are designed to disintegrate in upper part of Small Intestine.
It is available in BP/USP/NF/EP.
Applications :
1. Film coating of tablets
2. Enteric coating of tablets
3. Sustained release
4. Delayed release
5. Pallate coating material
Advantages of film coating :
▪️ Enhance palatability by masking unpleasant tastes or objectionable odours
▪️ Ease ingestion/swallowing
▪️ Improve product appearance
▪️ Protect tablets from light, oxidation & moisture
▪️ Advance the perception of superior product efficacy
-
Find Our Location
-
Call for Enquiries
-
Send Us a Message
About Us
G.M.Chemical started its operations in 1981 and has gradually emerged as not only the leading provider of Cellulose Acetate Phthalate [BP/USP/BPC/IP] to the Indian Pharmaceutical Industry but also to prestigious clients across the globe.
Quick Links
Products Link
Contact Us
- C-233/234 TTC Industrial Area, MIDC, Pawane, Navi Mumbai 400705, Maharashtra, India.
© 2026 G.M. Chemical. Designed by FQI. All rights reserved.